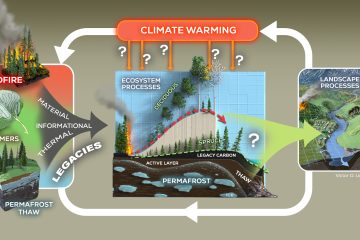
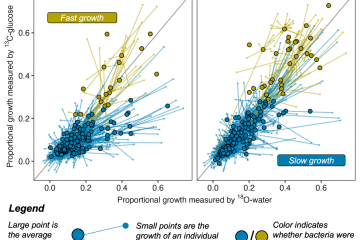
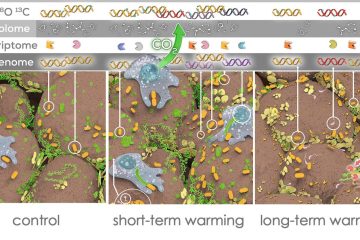
Probing carbon flux patterns through soil microbial metabolic networks using parallel position-specific tracer labeling
In order to study controls on metabolic processes in soils, we determined the dynamics of 13CO2 production from two position-specific 13C-labeled pyruvate isotopologues in the presence and absence of glucose, succinate, pine, and legume leaf litter, and under anaerobic conditions. We also compared 13CO2 production in soils along a semiarid substrate age gradient in Arizona. We observed that the C from the carboxyl group (C1) of pyruvate was lost as CO2 much faster than its other C atoms (C2,3). Addition of glucose, pine and legume leaf litter reduced the ratio between 13CO2 production from 1-13C pyruvate and 2,3-13C pyruvate (C1/C2,3 ratio), whereas anaerobic conditions increased this ratio. Young volcanic soils exhibited a lower C1/C2,3 ratio than older volcanic soils. We interpret a low C1/C2,3 ratio as an indication of increased Krebs cycle activity in response to carbon inputs, while the higher ratio implies a reduced Krebs cycle activity in response to anaerobic conditions. Succinate, a gluconeogenic substrate, reduced 13CO2 production from pyruvate to near zero, likely reflecting increased carbohydrate biosynthesis from Krebs cycle intermediates. The difference in 13CO2 production rate from pyruvate isotopologues disappeared 4–5 days after pyruvate addition, indicating that C positions were scrambled by ongoing soil microbial transformations. This work demonstrates that metabolic tracers such as pyruvate can be used to determine qualitative aspects of C flux patterns through metabolic pathways of soil microbial communities. Understanding the controls over metabolic processes in soil may improve our understanding of soil C cycling processes.