Dynamics of extracellular DNA decomposition and bacterial community composition in soil
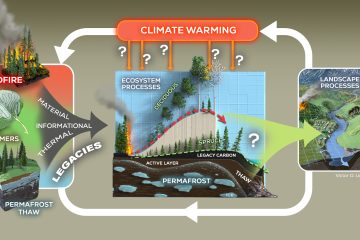
Microbial necromass is an important source of stabilized organic matter in soil, yet the decomposition dynamics of necromass constituents have not been adequately characterized. This includes DNA, a nutrient-rich molecule that when released into the environment as extracellular DNA (eDNA) can be readily used by soil microorganisms. However, the ecological relevance of eDNA as a nutrient source for soil microorganisms is relatively unknown. To address these deficits, we performed a laboratory experiment wherein soils were amended with 13C-labeled eDNA and clay minerals known to interact with DNA (kaolinite and montmorillonite). The amount of eDNA-carbon remaining in the soil declined exponentially over time. Kaolinite amendment decreased eDNA decomposition rates and, after 30 days, retained a higher fraction of eDNA-carbon (∼70% remaining) than control or montmorillonite soils (∼40% remaining), indicating that clay mineral sorption can stabilize eDNA-derived carbon in soil. Sequencing of bacterial 16S rRNA genes showed that during the incubation the relative abundance of the added eDNA’s sequence decreased by 98%, 92% and 99% in the control, montmorillonite, and kaolinite amended soils respectively. These results suggest that the fraction of eDNA-carbon that remained in the soil was incorporated into microbial biomass, firmly bound to soil constituents, or fragmented and no longer amenable to sequencing. In addition, the eDNA amendment affected the composition of the bacterial community. Specifically, the relative abundance of select phyla (Planctomycetes and TM7) and genera (e.g., Arthrobacter and Nocardioides) were elevated in soils that received eDNA, suggesting these groups may be particularly effective at degrading eDNA and using it for growth. Taken together, these results indicate that while eDNA is consumed by bacteria in soil, a fraction of eDNA material is resistant to decomposition, particularly when stabilized by soil minerals, suggesting a substantial amount of recalcitrant eDNA could accumulate over time.